전방척수동맥 침범을 동반한 척추동맥폐색에서의 혈관 내 치료: 두 증례의 상반된 임상 결과
Endovascular Treatment for Vertebral Artery Occlusion with Anterior Spinal Artery Involvement: Contrasting Outcomes in Two Cases
Article information
Trans Abstract
Vertebral artery (VA) occlusion is often managed conservatively, but anterior spinal artery (ASA) involvement may cause rapid neurological deterioration. We report two contrasting cases of VA occlusion with ASA compromise. One patient achieved complete recovery after timely endovascular treatment (EVT) restoring ASA flow, whereas the other developed severe bilateral medial medullary infarctions following unsuccessful EVT. These cases highlight the prognostic importance of ASA involvement and suggest that EVT should be considered when ASA perfusion is threatened.
Posterior circulation stroke caused by vertebral artery (VA) occlusion is generally managed conservatively when the basilar artery (BA) is not involved. However, VA occlusion affecting the anterior spinal artery (ASA) can lead to medullary infarction and rapid neurological deterioration, potentially resulting in catastrophic outcomes despite preserved BA flow.
Although advances in endovascular treatment (EVT) have expanded therapeutic options for posterior circulation strokes, the role of EVT in isolated VA occlusion remains unclear [1]. Importantly, ASA involvement is an underrecognized factor that may substantially alter the clinical course. Here, we report two contrasting cases of unilateral VA occlusion with ASA involvement: one with successful recanalization and complete recovery, and the other with failed revascularization and poor neurological outcome. These cases highlight the importance of early recognition of ASA compromise and its implications for treatment decisions.
CASE REPORT
1. Case 1
A 47-year-old man presented with sudden-onset headache 7 hours prior to admission and progressive right-sided weakness beginning 5 hours before arrival. He had a history of hypertension. Neurological examination revealed right hemiparesis, central-type facial palsy, right-sided hypesthesia, and dysarthria with a National Institutes of Health Stroke Scale (NIHSS) score of 6.
Magnetic resonance imaging (MRI) demonstrated acute infarction in the left medial medulla and V4 segment occlusion of the left VA (Fig. 1-A, B). Dual antiplatelet therapy was initiated. However, within 1 hour of arrival, the patient developed worsening hemiparesis with deterioration of the NIHSS score to 8, prompting the decision to pursue EVT.
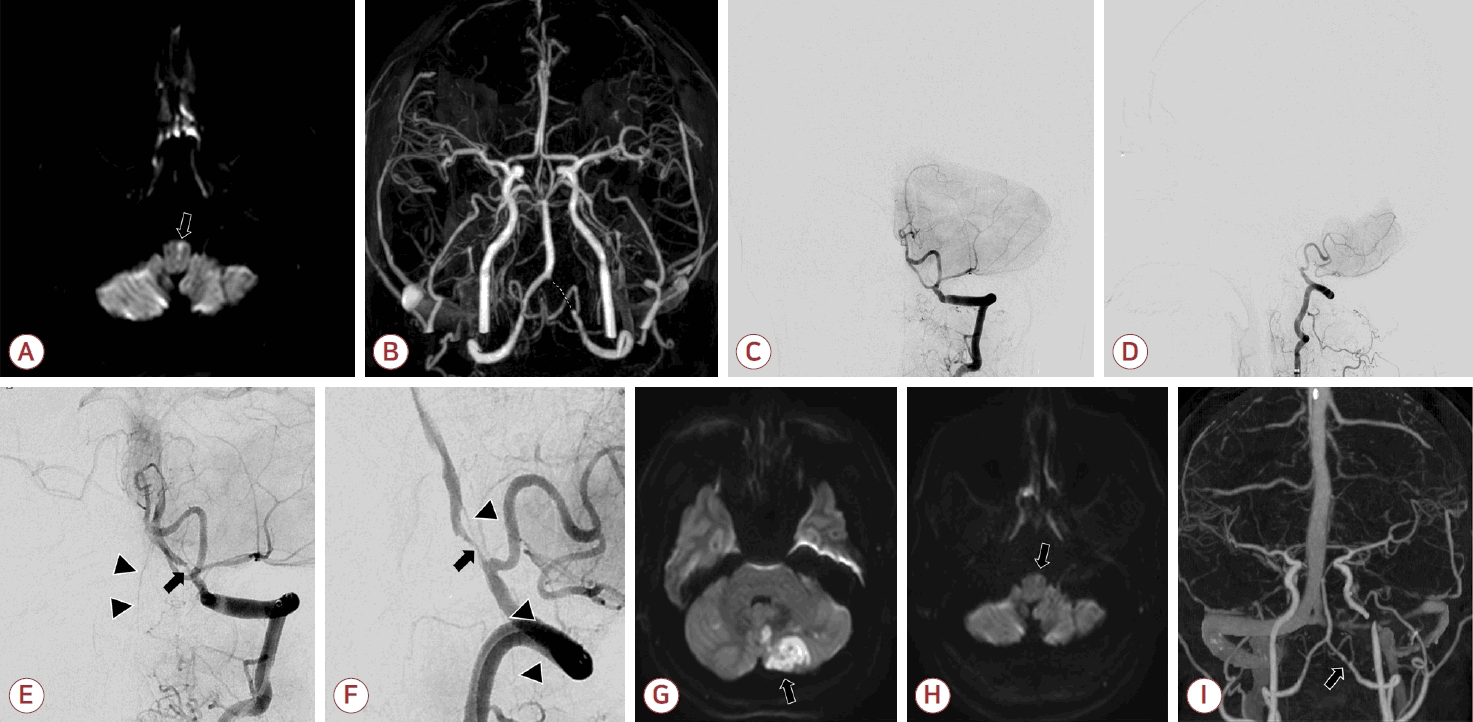
(A) Diffusion-weighted imaging (DWI) showing high signal intensity in the left medial medulla, indicating acute infarction (arrow). (B) Magnetic resonance angiography demonstrating occlusion of the left distal vertebral artery (VA) (dashed line). (C, D) Preprocedural vertebral angiography confirming VA occlusion distal to the posterior inferior cerebellar artery (PICA) origin: (C) frontal view and (D) lateral view. (E, F) After suction thrombectomy, vertebrobasilar flow was restored, and the anterior spinal artery arising from the vertebrobasilar junction became visible (arrowheads). However, severe residual stenosis remained in the left V4 segment (arrow). (G, H) Follow-up DWI demonstrated newly developed infarction in the left PICA territory (G, arrow), but decreased signal intensity in the medial medulla (H, arrow). (I) Computed tomography angiography obtained 6 months later showing residual stenosis of the VA but maintained patency (arrow).
Digital subtraction angiography confirmed left VA occlusion distal to the posterior inferior cerebellar artery (PICA) origin (Fig. 1-C, D). Suction thrombectomy using a 4-Fr 4Max Penumbra Aspiration Catheter (Penumbra, Alameda, CA, USA) achieved recanalization of left VA, but severe residual stenosis remained and re-occlusion occurred shortly thereafter. Repeat suction thrombectomy was performed, followed by intra-arterial tirofiban (2 mg) and a 12-hour intravenous infusion. Despite residual stenosis, anterograde VA flow was preserved, and post-recanalization angiography confirmed restoration of ASA flow arising from the left VA (Fig. 1-E, F).
Neurological deficits gradually improved. Follow-up MRI performed on the day after EVT revealed new cerebellar embolic infarcts, but decreased medullary lesion intensity (Fig. 1-G, H). The patient achieved complete recovery with a modified Rankin scale (mRS) score of 0 at 3 months. Follow-up computed tomography angiography at 6 months showed sustained patency of the left VA (Fig. 1-I).
2. Case 2
A 71-year-old man presented with acute dizziness 2 hours prior to admission. He had diabetes mellitus and atrial fibrillation. Neurological examination showed mild left-sided paresthesia (NIHSS score 1). Brain MRI demonstrated a small acute infarction in the right PICA territory, magnetic resonance angiography (MRA) revealed right VA occlusion (Fig. 2-A-C). Intravenous thrombolysis was not administered due to mild symptoms and apixaban was initiated.
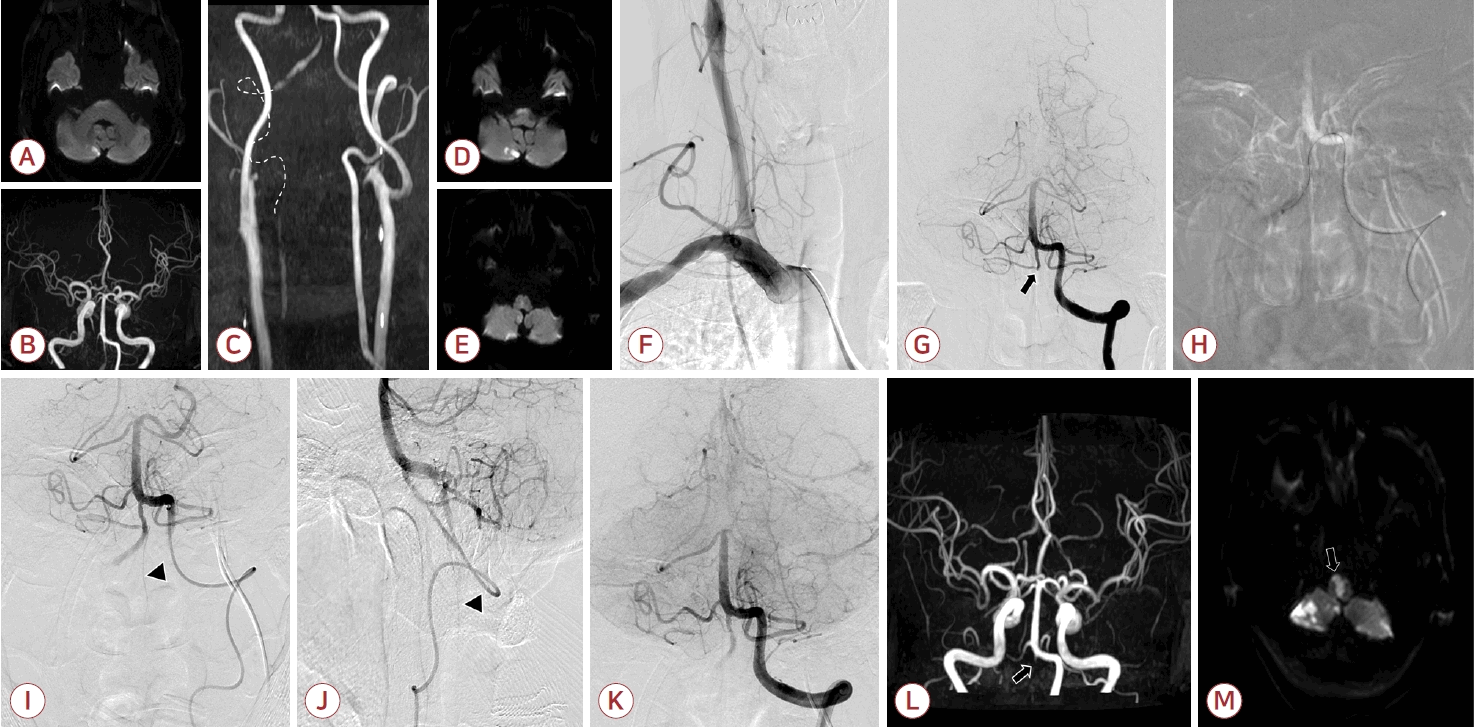
(A-C) Initial magnetic resonance imaging and magnetic resonance angiography (MRA) demonstrated focal cerebellar infarction with occlusion of the right V3 segment of the vertebral artery (VA) (dashed line). (D-F) Follow-up imaging on hospital day 1 after symptom worsening showed new acute infarction in the medulla on diffusion-weighted imaging (DWI). (F) Right subclavian angiography failed to visualize the VA origin. (G) Left vertebral angiography revealed occlusion of the right VA just distal to the posterior inferior cerebellar artery (PICA) origin (arrow), but no flow was identified at the V3-V4 junction that had been visualized on the initial MRA. (H) A suction catheter was advanced retrogradely from the left VA into the right VA, and suction thrombectomy was performed. (I, J) Partial recanalization of the right distal VA segment revealed anterograde flow in the anterior spinal artery (ASA) originating from the right VA (arrowhead). (K) During the procedure, repeated re-occlusion of the right VA resulted in loss of previously visualized ASA flow, and new occlusion of the right PICA was also noted. (L, M) Follow-up DWI revealed bilateral medial medullary and multiple cerebellar infarctions following failed recanalization of the right VA (arrow).
Approximately 9 hours after admission, neurological deterioration occurred, characterized by right-dominant quadriparesis and dysarthria, with the NIHSS score to 10, prompting follow-up MRI. Diffusion-weighted imaging demonstrated enlargement of the cerebellar infarction and newly developed high signal intensity in the dorsal medial medulla (Fig. 2-D, E). Based on these findings, emergent EVT for right VA occlusion was performed. Right subclavian angiography failed to visualize the VA origin, precluding anterograde access (Fig. 2-F). Left vertebral angiography showed partial retrograde filling of the right distal VA and PICA, but the V3-V4 junction, which had been seen on the initial MRA, was no longer visualized (Fig. 2-G). A retrograde approach via the left VA was attempted (Fig. 2-H). Suction thrombectomy using a 4-Fr AXS Vecta46 Catheter (Stryker, Portage, MI, USA) achieved partial recanalization up to the right V3 segment, with transient restoration of ASA flow that had not been visualized on the initial angiography (Fig. 2-I, J). However, repeated re-occlusion occurred, and final recanalization of the right VA was unsuccessful. Postprocedural left vertebral angiography demonstrated new occlusion of the previously patent right PICA, with no evidence of ASA flow arising from the left VA (Fig. 2-K). The patient’s neurological status rapidly worsened to drowsiness and quadriplegia (NIHSS score 16).
Follow-up MRI on hospital day 3 demonstrated extensive infarction involving the bilateral medial medulla and both cerebellar hemispheres (Fig. 2-L, M). Although mental status partially recovered, severe quadriparesis persisted. The patient was discharged to rehabilitation with an mRS score of 5 at 3 months.
DISCUSSION
Our cases demonstrate that clinical outcomes in VA occlusion can differ markedly depending on the success of reperfusion therapy, particularly when the ASA is compromised. These contrasting outcomes suggest that VA occlusion with ASA involvement represents a distinct and potentially life-threatening clinical entity, in which EVT may be required in selected cases.
A recent case series has proposed that neurological deterioration in patients with unilateral VA occlusion and patent BA may result from ischemia of VA perforators, and that EVT may prevent further clinical worsening [2]. This concept aligns with the rapid neurological deterioration observed in our patients. While a previous report inferred perforator ischemia based on clinical presentation and infarct patterns, our cases add supportive evidence by demonstrating angiographic involvement of the ASA and its dynamic changes before and after EVT.
The ASA supplies critical structures in the medial medulla and upper cervical spinal cord, including the pyramidal tract, medial lemniscus, hypoglossal nucleus [3]. ASA occlusion can even result in bilateral medial medulla infarction, resulting in rapidly progressive deficits such as hemiparesis or quadriparesis, bulbar symptoms, and respiratory compromise [4,5]. Importantly, motor weakness is uncommon in isolated VA occlusion unless VA perforators or the ASA are involved. Thus, the presence or progression of motor deficits should be regarded as a clinical red flag suggesting ASA compromise.
A common misconception is that ASA infarction requires bilateral VA occlusion, as the ASA is traditionally depicted as arising from the confluence of both VAs. However, anatomical studies have demonstrated substantial variability, including unilateral origin or asymmetric contributions [6-8]. Such variants explain how unilateral VA occlusion can precipitate ASA territory infarction, as seen in our cases. Failure to recognize this variability may lead to underestimation of the risk associated with isolated VA occlusion.
The divergent outcomes in our patients underscore the importance of timely and effective reperfusion. In the first case, early recognition and successful EVT restored ASA perfusion, resulting in complete recovery. In contrast, failure to maintain VA patency in the second case led to persistent ASA hypoperfusion and devastating bilateral medial medullary infarction. These finding suggest that when ASA involvement is suspected, aggressive EVT aimed at restoring and maintaining ASA flow may be warranted, even in unilateral VA occlusion.
In conclusion, unilateral isolated VA occlusion should not be presumed benign when motor weakness or progressive neurological symptoms are present, as these findings may indicate ASA compromise. Given the marked anatomical variability of ASA, infarction can occur even with a single VA occlusion. Early recognition of ASA involvement and timely, successful EVT may be crucial to prevent irreversible neurological deficits in this subset of posterior circulation strokes.